- SURGICAL
Latest Technologies
- Areola and Nipple Correction
- Breast Augmentation
- Breast Augmentation with Fat Transfer (No Implants)
- Breast Lifts
- The Au Naturel Signature Breast Lift
- Tuberous Breast Repair
- Benelli Breast Repair
- Male Gynecomastia
- Breast Revision
- Breast Reduction
- Breast Implant Correction
- Breast
- Scarless Breast Reduction
- Tummy Tuck
- Liposuction & Liposculpting
- Renuvion
- Natural Brazilian Butt Lift
- Mommy Makeover
- Belly Button (Umbilicoplasty)
- Revision Surgery/Liposuction Revision
- C-Section Scar
- Thigh Lift
- Body Lift
- Arm Lift
- Arm Liposuction
- Male liposuction
- Body
- Cellulite
- Aveli Cellulite Treatment
- Cellulaze™ for Los Angeles
- NON-SURGICAL
- Cellulite & Fitness Treatments
- Emsculpt®
- Emsculpt Neo
- Cellulaze™ for Los Angeles
- Wrinkle Relaxers
- Dysport® XC
- Jeuveau
- Botox®
- Dermal Fillers
- Skinvive™
- Juvéderm Voluma®
- Juvéderm® XC
- Radiesse®
- Restylane®
- Restylane® Lyft
- Restylane® Silk
- Other Procedures
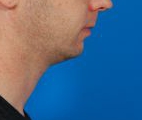
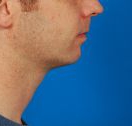
- Kybella®
- Eyelash Enhancement
- Sculptra Aesthetic®
- Sclerotherapy Vein Treatment
- Upneeq Eye Drops
- Skin Rejuvenation
- Led Light Therapy
- Facial Thread Lift
- ABOUT
- PHOTOS
- FEATURED CASE STUDIES
- RESOURCES
- CONTACT